Abstract
India's product information systems were built to answer admissibility questions - is this product safe? Can it cross this border? They answer these questions correctly. They were never designed to answer intelligibility questions - what is this specific product, by whom was it grown, what are its attributes?
This synthesis paper identifies the mechanism that produces this gap - the non-accumulation mechanism - and maps it across eight information domains. It examines why India has already solved this problem once (TraceNet), why that solution was never generalised, and what structural conditions any future solution must satisfy. The paper extends the analysis beyond food to textiles, cosmetics, and pharmaceuticals, where the same mechanism operates through different regulatory gateways.
The paper does not prescribe a solution. It defines the structural conditions that any solution would need to satisfy, and identifies the governance question that must be answered before those conditions can be met.
STRUCTURAL GAPS IN PRODUCT INFORMATION SYSTEMS SGPIS-SY · Synthesis
What the Product Knows: India's Missing Intelligence Layer
Shams Ahmed Altibbe Inc. April 2026
Altibbe Research
The farmer knows the variety, the soil, the practice, the season — exactly what any serious buyer needs to evaluate the product. Between them sits a documentation stack — phytosanitary certificate, HS commodity code, certificate of origin, laboratory test result — that establishes whether the product may cross a border and answers nothing about the product itself. The buyer asks what it is. The documentation says it is safe to import. The farmer's knowledge does not travel.
This paper is about that gap — where it appears, why it persists, and what the structural conditions for closing it are.
EXECUTIVE SUMMARY
India's product information systems were built to answer the right questions for their era. Is this product safe? Does it meet the threshold? Can it cross this border? These are admissibility questions, and the systems built to answer them — FSSAI licensing, export certification, commodity grading, geographical indication registration, traceability platforms — answer them correctly. They were designed well. The fact that they continue to function is testimony to how well they were built.
What has changed is not the validity of those systems. It is the speed, frequency, granularity, and multiplicity of uses now expected from product-linked information. A domestic health claim submission requires producer-level nutritional composition with documented methodology. An international buyer's due diligence requires origin specificity, practice documentation, and supply chain evidence. A premium market positioning requires the story of a specific product from a specific producer with specific attributes. These are intelligibility requirements — and the inherited admissibility architecture was never built to carry them.
The gap between what Indian producers know about their products and what their documentation systems can communicate is not a failure of any actor or institution. It is an inherited structural condition. Systems built to establish admissibility under earlier technological and institutional constraints are now being asked to support forms of intelligibility, portability, and reusability they were never required to carry at the time of their creation.
This paper identifies the mechanism that produces this condition — the non-accumulation mechanism — and maps it across the domains in which it operates. The mechanism works in three steps: information is generated within bounded institutional purposes, isolated by system-specific schemas, and reconstructed rather than carried at every institutional boundary. The distinctive feature is not that information sits in separate systems. It is that each system was built with no forward requirement, making reconstruction at the next boundary structurally rational rather than incidental.
The mechanism persists because of three reinforcing factors: no institution owns the connective governance question, liability structures disincentivise specific declaration in the absence of supporting infrastructure, and workflow configurations have embedded reconstruction as standard practice. Its scope is precisely delimited — it is the binding constraint for input-based compliance requirements (traceability, health claim substantiation, sustainability due diligence) and does not operate in the same way for output-based threshold requirements.
India has demonstrated, within one bounded context, that the mechanism can be overcome. TraceNet, administered by APEDA for organic exports, captures structured, producer-attributed declarations and has operated for two decades. It is not connected to BharatTradeNet, BAN Lab Setu, or ONDC. The constraint that prevented generalisation was never technical. It was the absence of a mandate to extend the model beyond the corridor for which it was specifically built.
Among the consequences of this structural condition, the health information gap carries one of the most direct human implications. When producer-level nutritional and composition intelligence cannot travel to consumers, regulators, and buyers in structured portable form, health claims cannot be substantiated at source, dietary choices cannot be informed at point of purchase, and the food safety framework cannot see upstream of the threshold it is designed to enforce.
This is not only India's inherited condition. Every trading economy built the same admissibility architecture under the same constraints. The structural conditions that produced the gap are present wherever producer knowledge has acquired downstream value — and the governance question this paper identifies is not specific to India. It is the question every economy faces when its admissibility architecture is asked to carry intelligibility requirements it was not designed to satisfy. This paper does not prescribe a solution. It identifies the structural conditions that any solution would need to satisfy and the governance question that must be answered before those conditions can be met.
1. THE SITUATION
1.1 The Domestic Policy Convergence
India's food policy institutions are not standing still. The Supreme Court, in 3S And Our Health Society v. Union of India & Anr., has directed FSSAI toward concrete progress on front-of-pack nutrition labelling, finding that existing measures have not yielded adequate results and that the constitutional right to health requires action.⁴ FSSAI's structured submission requirement for health and nutrition claims, effective January 1, 2026, has opened a structured evidentiary gateway — health claims must now be substantiated with producer-specific nutritional composition data, documented methodology, and India-specific consumption context.³ The Digital Agriculture Mission has integrated over 84 million farmer identifiers into a federated digital infrastructure with interoperability design principles.
The Jan Vishwas (Amendment of Provisions) Act, 2026, passed by both Houses of Parliament in April 2026, shifts certain procedural violations in food and export statutes from criminal to civil penalties.¹⁵ᵇ It signals a legislative direction toward frameworks that assume producer good faith — a direction whose value increases in proportion to the quality and specificity of the declarations producers are able to make.
BharatTradeNet is being designed to unify India's trade documentation infrastructure. APEDA runs buyer-seller meets for millets — the crop India produces in greater quantity than any country on earth. Each of these initiatives is real. Each reflects genuine institutional commitment. And each, in its design, assumes something this paper will show is not yet true: that what Indian producers know about their specific products exists in a form that can travel — to the regulator reviewing a health claim, the buyer evaluating an export consignment, the consumer deciding at the point of purchase.
1.2 The External Pressure
From the export end, a distinct set of pressures is generating equivalent demand for producer-level product information.
The EU Deforestation Regulation establishes legally binding requirements for producer-linked, plot-traceable documentation as a condition of EU market access for covered commodity categories.⁷ India's low-risk classification simplifies but does not eliminate due diligence obligations: Article 9 information collection — including plot-level geolocation — remains required regardless of risk classification.⁹ Coffee Board of India enrolment drives had reached approximately 2% of India's coffee growers in EUDR compliance systems as of December 2025, even with a dedicated government application and institutional support.³²
Beyond public regulation, private buyer standards and retailer codes of practice impose equivalent upstream intelligence requirements. GlobalGAP certification, BRCGS food safety standards, and major retailer supplier protocols each require structured documentation of practices, origin specificity, and product attributes that the admissibility stack does not carry. These requirements are currently satisfied through bespoke bilateral audits: expensive, non-portable, and reset with every new buyer relationship. They partially solve the intelligibility problem for those who can afford repeated reconstruction. They do not solve it systematically, and they confirm that the mechanism applies to private and public documentation regimes equally.
At a reverse buyer-seller meet under the RAMP programme in Hyderabad in April 2026, a European buyer sourcing rice made his commercial sourcing decision based on cooking a product sample at home.¹⁰ The standard export documentation did precisely what it was designed to do: it established admissibility. It was never designed to answer the buyer's question about product differentiation. The producer knew what the buyer needed. Nothing in the documentation architecture was designed to carry it.
The US Food Safety Modernization Act's Foreign Supplier Verification Program requires US importers to verify that foreign suppliers can demonstrate process-level safety controls — not only output-level test results. The UAE requires a Free Sale Certificate for food products for which no Indian national equivalent exists. Across destination markets, what varies is the specific requirement. What is consistent is the direction: both public regulation and private buyer standards are shifting toward producer input data that India's documentation architecture was not built to generate.
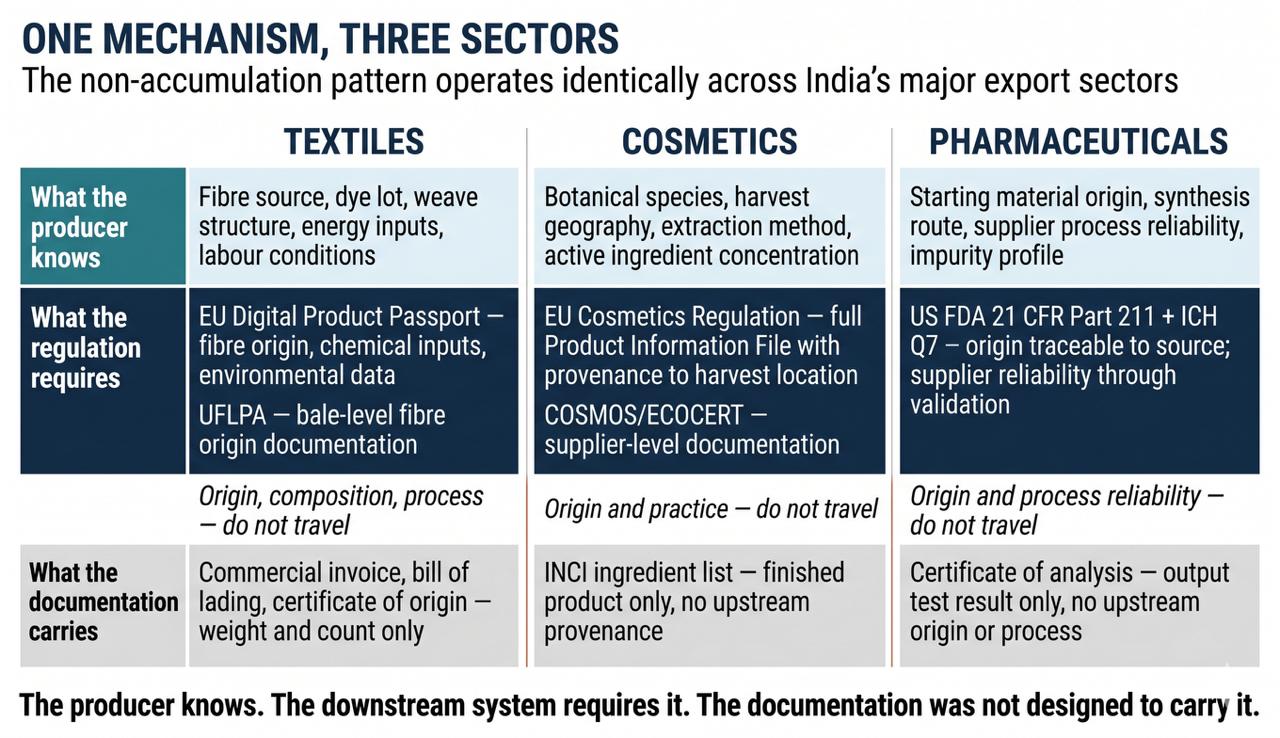
The same structural pressure is operating in parallel across non-food sectors. The EU's Ecodesign for Sustainable Products Regulation will require Digital Product Passports for textiles by 2030 — product-level records carrying fibre composition, chemical inputs, and environmental data from producer to consumer.⁴⁰ The US Uyghur Forced Labor Prevention Act creates a rebuttable presumption of forced labour for goods with supply chain connections to Xinjiang, requiring Indian textile exporters to document fibre origin at bale level; cotton and textiles account for over 60% of UFLPA detentions.⁴¹ The EU Cosmetics Regulation (EC) No 1223/2009 requires a full Product Information File for botanical and herbal ingredients — documenting extraction solvent, drug-extract ratio, and botanical source — that Indian ayurvedic and personal care exporters currently satisfy through bilateral audits that reset with every buyer relationship.⁴² US FDA 21 CFR Part 211 and ICH Q7 require origin documentation traceable to source for every API starting material; Indian generic pharmaceutical manufacturers, supplying approximately 20% of global generic exports, face this requirement as a current enforcement reality with FDA warning letters and import alerts directly citing supplier-reliability documentation failures.⁴³ In each of these sectors, the mechanism is identical: the producer holds the knowledge, the downstream system requires it, and the documentation architecture was not designed to carry it in portable form.
1.3 The Shared Assumption
The domestic disclosure agenda and the export documentation agenda are institutionally separate. They originate from different regulatory traditions, operate through different agencies, and serve different policy objectives. They share one structural assumption that neither has yet built the infrastructure to satisfy: that what Indian producers know about their specific products exists in a form that can travel across institutional boundaries without reconstruction.
That assumption is not unique to food. It is present wherever what a producer knows about their product determines what a buyer will pay for it — in textiles, in cosmetics, in pharmaceuticals, in handicrafts. What makes food systems the entry point for this analysis is the convergence of simultaneous domestic and international pressures that makes the gap most visible and most immediately consequential here. The structural condition is the same across sectors. The paper traces it through food systems because that is where the evidence is most fully developed and where the policy moment is most acute.
That assumption is the subject of this paper.
2. WHERE THE INFORMATION DISAPPEARS
2.1 What Has Been Built
Look at what India has actually constructed. Eighty-four million farmers with digital identities in AgriStack, incorporating land records and crop sown data. Fertiliser purchase records tied to individual farmer identities through the Direct Benefit Transfer system. Laboratory test results digitised and QR-verified through BAN Lab Setu. Organic certification records with farmer-level practice data through TraceNet. FSSAI licensing covering 1.2 crore food business operators.
Each of these systems works. Each was built at significant cost and with serious institutional effort. The question is not whether India has built information infrastructure. It clearly has. The question is why none of it accumulates into something a buyer in Hamburg can access when they want to know where their turmeric came from, how it was grown, and what its curcumin concentration is.
2.2 What Each System Was Built to Answer
Each system was built to answer a specific institutional question, and each answers it correctly. AgriStack answers whether a farmer is eligible for scheme benefits. BAN Lab Setu answers whether a consignment has passed the required laboratory tests. FSSAI licensing answers whether a food business operator is registered and compliant. Each is an admissibility system — designed to generate a determination about whether a subject meets a defined threshold for a defined purpose. The systems are not failing. They are doing precisely what they were designed to do.
The problem is that the question none of them was built to answer — what does this specific producer know about this specific product, and how does that knowledge travel to the institutions and buyers who need it — is precisely the question that India's current policy moment is now asking.
2.3 The Moment the Information Disappears
It disappears at every handoff.
The farmer knows the variety. When they sell to the aggregator, the variety does not travel — the aggregator buys by grade. When the aggregator sells to the exporter, the grade does not travel — the exporter packages by specification. When the exporter submits to APEDA, the specification does not travel — what travels is the HS code. When the HS code reaches the buyer, they know India exported a commodity. They do not know what the farmer grew.
At each of those handoffs, documentation service providers perform a necessary and legitimate bridging function: gathering and reformatting product information to meet the requirements of each successive system. This reconstruction is standard practice across the export chain. It is a rational response to an architecture in which information generated for one purpose cannot travel to the next in the form the next system requires. The reconstruction exists because the architecture makes it necessary — not because any actor within it is performing their role incorrectly.
This is the non-accumulation mechanism. The distinctive feature is not that information sits in separate systems — it is that each system was built with no forward requirement, making reconstruction at the next boundary structurally rational rather than incidental. Information is generated at every stage, but at every stage it was generated for one purpose and cannot cross to the next purpose without being rebuilt from scratch. Nothing accumulates into portable product identity. The consequence is a compounding cost structure that falls most heavily on smaller producers. For a large organised exporter, documentation reconstruction is a fixed overhead distributed across transactions. For a smallholder or mid-sized producer, it is a variable cost that resets with every new buyer, every new corridor, every new compliance requirement — concentrating the access barrier precisely where the most differentiated and attributable products originate.
2.4 Why the Gap Persists
After more than a decade of active digital infrastructure investment — systems that work, institutions that function, officials who are committed — the information still does not travel. Three structural factors keep it in place.
The governance condition. India's apex institutions have acknowledged the fragmentation explicitly. NITI Aayog's 2025 agricultural technology assessment states that agriculture data "remains fragmented and siloed, with no single source of truth."ᴺ¹ The Economic Advisory Council to the Prime Minister concluded in 2022 that "a streamlined information-sharing mechanism needs to be put in place" across 17 responsible ministries — implicitly acknowledging that none currently exists.ᴱ³ A parliamentary standing committee recorded that government data on farmers "were in different silos — not coherent or usable."ᴾ⁷ Each acknowledgment is made from within a single institutional vantage point. None names the cross-domain question — how producer-level product information should be structured and carried across systems so that it satisfies FSSAI's requirements, APEDA's documentation standards, and international buyer due diligence from a single producer declaration — as a discrete governance responsibility to be assigned. The fragmentation is seen. The question of who should govern the connective layer is not posed.
Legal and institutional rationality. A threshold determination assigns clear accountability and creates a defined safe harbour. A granular producer declaration — specific variety, measured concentrations, documented method — creates specific liability. Specificity is actionable. Generality is survivable. Under India's food safety statute, producers and exporters face concrete incentives to confine their representations to what threshold compliance frameworks can verify. Granular declaration is not only technically unsupported — it is, in the absence of infrastructure that makes it accurate and reusable, institutionally cautious. The liability structure did not produce the admissibility architecture; but it reinforces it.
Workflow entrenchment. Export documentation crosses from producer through aggregator, processor, exporter, pre-shipment inspection body, certification body, regulatory interface, and customs. At each transition, product information is rebuilt for the next system's requirements. Single Window systems reduce submission friction but do not eliminate duplicate data entry — each agency continues to operate its own schema. The reconstruction pattern is structurally reinforced by the configuration of actors, not merely by absent good practice.
2.5 The Scope Qualifier
The non-accumulation constraint does not apply uniformly. Chile achieved over 93% industry compliance with mandatory front-of-pack nutrition labelling without requiring any centralised producer data infrastructure.²⁴ Threshold labelling is output-based compliance — it asks whether a finished product exceeds a defined nutrient level, a determination made at the product output stage without reference to producer-level input data. Non-accumulation does not impede output-based compliance because output-based compliance does not require information to travel from the point of production.
It is also true that for certain products at the high end of the premium market, intelligibility is currently achieved through direct buyer relationships, bilateral audits, and private certification schemes. These mechanisms partially solve the problem — expensively, bilaterally, and without the portability that would make the solution reusable across buyers and corridors. They do not solve it systematically, and they do not serve the transition from admissible commodity to attributable premium product that India's current policy agenda is attempting at scale. The NSC portal, the millet export mission, the GI premium capture agenda, and the trust-based governance shift all target precisely this transition. The non-accumulation mechanism is the binding structural constraint at exactly this point.
The category of requirement that is growing — across India's domestic disclosure trajectory and its international market access environment simultaneously — is input-based compliance. Traceability requires documented production origin and chain of custody. Health claim substantiation requires producer-level composition with documented methodology. Sustainability due diligence requires evidence of how a product was produced. These cannot be satisfied by output-stage determination.
2.6 The One Place It Has Worked
India has already solved this problem once. TraceNet, administered by APEDA for organic exports, captures farmer identity, crop variety, GPS coordinates, yield estimates, and input use records through a formal Internal Control System built on grower groups of 25–500 farmers. The data is structured, attributable, and used for regulatory purposes across approximately two decades of operation.
TraceNet was built because a specific external mandate — EU organic equivalency recognition — compelled one agency to build one corridor-specific system. That mandate created the infrastructure. The same infrastructure is not connected to BharatTradeNet, BAN Lab Setu, or ONDC. For EU TRACES NT submission, Indian organic exporters re-enter data separately — no interoperability mechanism exists despite both systems serving the same export corridor. The model has not been extended beyond organic exports not because it failed, but because no institution has been assigned the mandate to extend it.
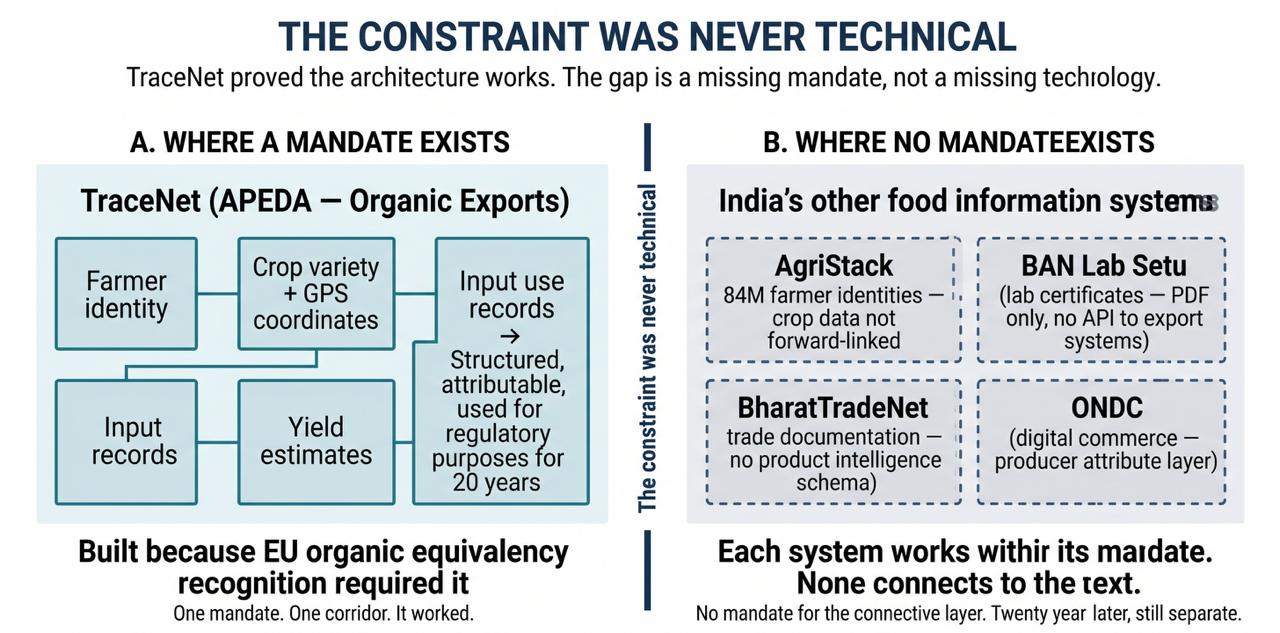
“TraceNet is not an exception to the mechanism. It is the mechanism’s clearest confirmation: proof of what becomes possible when a mandate exists, and proof of what persists when the mandate stops.”
This is the paper's most important single finding: India has already built, once, exactly the kind of producer-level declaration infrastructure the mechanism predicts cannot exist. It worked. The constraint was never technical. It was the absence of a mandate to generalise the model beyond the corridor for which it was specifically designed. This pattern holds across every system examined in this research: where producer-level information portability has been achieved, it operates within a bounded mandate — a specific commodity, certification type, or export corridor — and does not extend beyond it. No system examined has achieved cross-purpose portability without an explicit institutional mandate. TraceNet is not an exception to the mechanism. It is the mechanism's clearest confirmation: proof of what becomes possible when a mandate exists, and proof of what persists when the mandate stops.
2.7 The International Dimension
India's documentation systems establish that a product is safe for export. Importing systems — across EU, US, UAE, and GCC regulatory frameworks, and across the private standards of major retail and institutional buyers — increasingly ask how it was made safe: by whom, under what conditions, with what inputs, to what measured specifications.
The gap is not uniform across regimes, and requirements vary. What is consistent is the direction: documentation expectations are shifting toward producer input data that India's admissibility architecture was not built to generate. The gap is not a matter of additional documents in the same system. It is a structural incompatibility between documentation layers reproduced at every institutional and commercial boundary.
3. THE TERRITORY
The non-accumulation mechanism is not confined to a single corridor or domain. It recurs across the information landscape of India's food system — and, beyond food, across every sector in which what a producer knows about their product has value to a downstream actor that the current documentation architecture cannot carry. Food systems are the entry point for this analysis because the convergence of domestic disclosure pressure and international market access requirements makes the gap most visible and most urgent here.
The following table maps the mechanism across eight information domains, organised into four clusters. Rows directly evidenced by named systems and official assessments carry the strongest evidentiary weight. The remaining rows reflect documented structural observations. The table is a map of where the mechanism recurs, not a uniform empirical claim of equal weight across all rows.
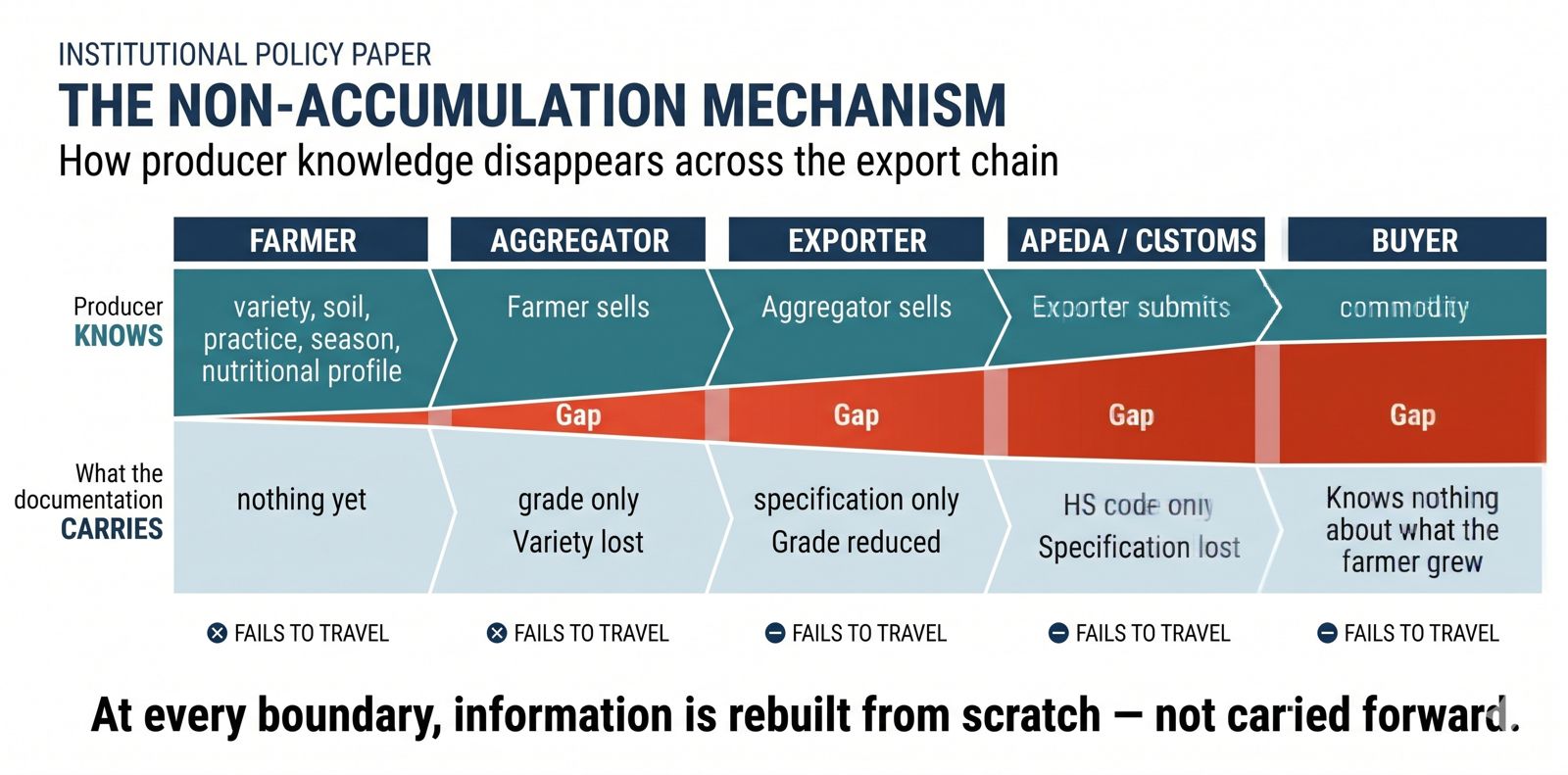
Table 1: The Non-Accumulation Mechanism Across Information Domains
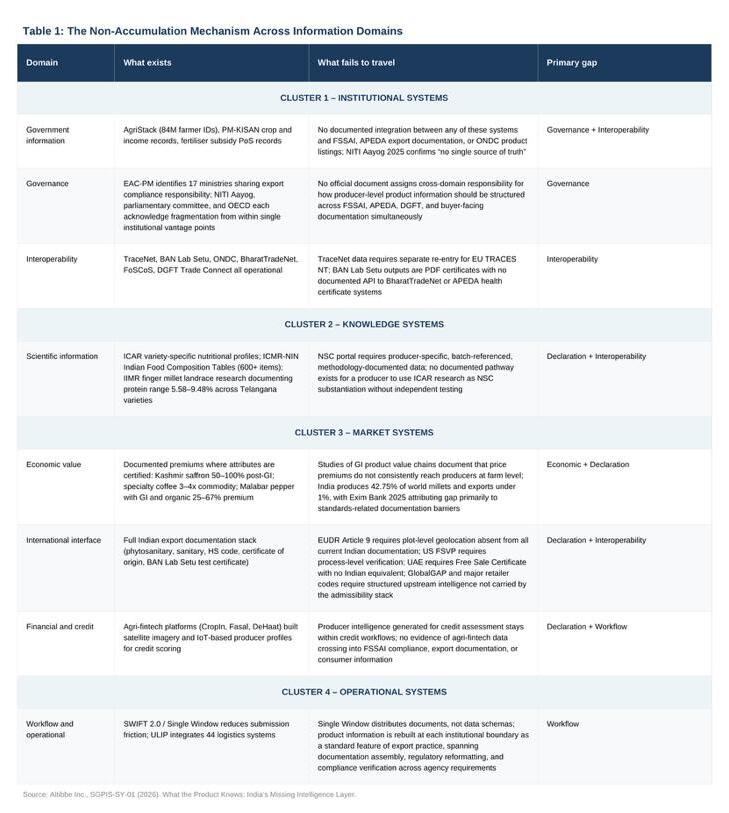
The pattern across clusters is consistent: information generated within bounded institutional purposes does not accumulate into portable product identity. The mechanism is not a feature of one domain. It is a property of how the architecture was built.
The same property operates across sectors beyond food. An Indian textile manufacturer in Tiruppur knows the fibre source, the dye lot composition, the weave structure, and the energy inputs of every production run. A European buyer requiring Digital Product Passport compliance under the EU Ecodesign Regulation needs exactly this information in structured portable form.⁴⁰ The documentation architecture between them — commercial invoice, bill of lading, certificate of origin, packing list — carries weight and count, not origin, composition, or process. A European Parliament STOA study published in July 2024 found that textile supply chains continue to collect producer-level data "in the conventional way using Excel sheets and manually filling in documents," with fibre-level traceability identified as the critical gap.⁴⁴ UFLPA detentions carry costs exceeding USD 810,000 per case, driven primarily by inability to prove fibre origin within CBP's 30-day compliance window.⁴¹ An Indian ayurvedic cosmetics producer knows the botanical species, harvest geography, extraction method, and concentration of every active ingredient. The EU Cosmetics Regulation requires a full Product Information File anchored to this knowledge.⁴² The documentation architecture carries the finished product's INCI ingredient list — not the producer's upstream knowledge that makes provenance verifiable. The consequences are measurable: DNA-barcoding studies published in 3 Biotech document that 20.4% of commercial ashwagandha root extract samples contained undisclosed aerial-part substitution, attributable to absent producer-level provenance documentation in the supply chain.⁴⁵ An Indian API manufacturer knows supplier process reliability, starting material origin, and incoming material variation accumulated through years of supplier interaction. ICH Q7 and 21 CFR Part 211 require this knowledge in documented, auditable form.⁴³ The FDA's February 2025 warning letter to Tyche Industries Ltd. cites the gap in regulatory-enforcement language: the firm relied on supplier certificates of analysis without establishing supplier reliability through validation, and the regulator required that supplier reliability be established "through initial validation as well as periodic re-validation" — evidence of a knowledge state the documentation schema does not operationalise.⁴⁶ Indian pharmaceutical manufacturers face repeated FDA enforcement actions, with documentation-related citations — inadequate supplier qualification, unverified raw material origin, incomplete audit trails — consistently among the most cited categories in warning letters and import alerts to Indian sites. In each case: the producer knows. The downstream actor needs. The architecture between them was not designed to carry it.

4. WHAT THIS LOOKS LIKE ON THE GROUND
4.1 The Farmer Who Knows Everything
A smallholder in Nalgonda district grows finger millet — not generic finger millet, but a local Rajapore landrace that IIMR research has documented carrying protein content between 7.2% and 9.1%, significantly above the category average, with calcium concentration of approximately 331 milligrams per 100 grams — nearly ten times that of wheat or rice.²⁶ The farmer has cultivated this variety on this land for twenty years. They know the soil, the seasonal variation, the harvest timing that produces the highest nutritional concentration.
They want two things. Domestically, they want to label their product as a good source of calcium and high in dietary fibre — claims that FSSAI's own millet guidance identifies as scientifically supportable for the category. To substantiate those claims through the NSC portal, they need producer-specific nutritional composition data with documented methodology and India-specific consumption context. The IIMR research exists. The ICMR-NIN tables exist. But neither constitutes the producer-specific, batch-referenced, methodology-documented data the NSC portal requires. The farmer must commission independent testing they cannot afford, to substantiate a claim about a nutritional fact that government-funded research has already established for their variety. The claim is true. The infrastructure to carry it to the portal does not exist at their scale.
From the export end: India produces 42.75% of the world's millets and exports less than 1% of that production.²⁸ The Exim Bank's 2025 assessment identifies standards-related documentation barriers as the primary obstacle to growth in premium export markets.²⁷ A buyer in Amsterdam will pay more for documented variety, documented origin, documented nutritional profile, documented cultivation practice. The Nalgonda farmer has all of that knowledge. The documentation system carries none of it.
The same gap is visible from both directions simultaneously. The domestic compliance architecture and the export documentation architecture each require the same missing layer. Neither was designed to generate it.
4.2 The Manufacturer Between Two Requirements
A mid-sized processed food manufacturer produces a multigrain functional snack. The product contains millets, pulses, and traditional spice blends in proportions chosen for specific nutritional outcomes — elevated protein, high dietary fibre, reduced glycaemic index. The manufacturer wants to carry health claims on the domestic label. The NSC portal, effective January 2026, requires producer-specific nutritional composition data for each ingredient, with documented methodology, batch references, and India-specific consumption evidence.
The data exists — in procurement records, supplier agreements, and internal quality assurance testing. But it exists in separate repositories, at different levels of specificity, held by different functions within the organisation, and not in the standardised structured format the NSC portal requires. Assembling it into a compliant dossier requires significant effort and specialist knowledge. A large food company with a dedicated regulatory affairs team absorbs this as overhead. This manufacturer, with a lean team focused on production and sales, faces a barrier that has nothing to do with the quality of their product or the truth of the claims they want to make.
From the export end, the same manufacturer has received interest from a European buyer seeking a certified multigrain blend meeting BRCGS Grade A standards, with full traceability to ingredient source and documented nutritional composition per batch. BRCGS Grade A requires a comprehensive food safety management system, documented hazard analysis, and supply chain verification extending to primary ingredient producers. The manufacturer's ingredients are sourced from known suppliers; the traceability information exists informally. It does not exist in the structured, auditable, portable format that BRCGS certification requires as a precondition of supply.
The same gap appears from both ends. The domestic health claim and the international buyer requirement each demand the same structured upstream producer intelligence. The manufacturer holds it in fragments across internal systems. The documentation infrastructure was not designed to make it portable, and the gap is structural — not a consequence of the manufacturer's size or capability.
4.3 The Heritage Product Without a Documentation Pathway
India's Geographical Indication registry protects 658 products. The GI system was designed to protect the relationship between a product name and its place of collective origin. It performs that function correctly.
Within every GI zone, individual producer variation remains outside the GI record. A Darjeeling tea producer whose garden sits at a specific altitude, uses a specific cultivar, and follows a specific orthodox processing method produces a product whose attributes are demonstrably distinct from other Darjeeling teas within the same zone. The buyer who would pay a premium for those specific attributes cannot access the documentation to verify them. The GI establishes the collective boundary. It does not describe what lies within it at the producer level.
The premium market for geographically specific, attribute-documented food products is real, growing, and actively seeking Indian products. The GI protects the name. The market wants what lies within it — the producer, the practice, the specific attributes that make one Darjeeling different from another. The documentation infrastructure connects neither.
5. WHAT THIS MEANS
India's active food policy initiatives — FOPL, health claim substantiation, Jan Vishwas, BharatTradeNet, the millet export mission, AgriStack — are each running into the same missing layer. Not because they are badly designed. Because they were designed downstream of a condition they assumed exists but does not yet.
For food safety and disclosure regulators: The NSC portal's structured submission requirement will generate compliance asymmetry as a stable outcome, not a transitional one. Large food business operators with in-house scientific capacity can generate, structure, and submit producer-specific nutritional composition data. Smaller producers — who represent the majority of India's food producer base and often carry the most differentiated traditional and regional attributes — cannot. Twenty years of EU health claims regulation produced evidence that substantiation requirements, without upstream information infrastructure, function as barriers to entry rather than quality standards.²² The front-of-pack label format decision — tabular or pictorial — resolves a downstream design question while leaving the upstream condition unaddressed. Chile's 93% FOPL compliance was not produced by superior label design. It was produced by a mandatory producer data obligation that created the upstream pressure the downstream label required.²⁴
Of the consequences of the structural condition, health carries one of the most direct human implications. When producer-level nutritional and composition intelligence cannot reach consumers in structured portable form, informed dietary choice is not possible at the scale the front-of-pack architecture assumes.
For export promotion and trade agencies: The buyer-seller meets will continue. The market development work will continue. And at each meet, buyers will ask questions — where exactly, how exactly, by whom exactly — that the documentation stack the exporter brings cannot answer. The gap between what buyers need to know and what India's documentation can carry is not narrowing. It is widening as both public regulatory requirements and private buyer standards move faster than documentation architecture. Export promotion operates on the demand side of a documentation gap that falls outside any export agency's current mandate.
For digital agriculture and infrastructure institutions: AgriStack's design principles — open APIs, national portability, interoperability intent — are exactly right. The Farmer ID is a genuine achievement. And yet the system that has captured more structured producer-level data than any other — TraceNet — is not integrated with AgriStack, BharatTradeNet, or BAN Lab Setu. The infrastructure exists in parallel rather than in connection. The connective layer between what each system captures and what the next system needs remains unbuilt — not because the technology is insufficient, but because no institution has been assigned the governance mandate to build it.
For producers whose products carry more value than their documentation can communicate: The asymmetry compounds. Large organised producers build documentation capacity as a fixed cost distributed across transactions. Smaller producers face documentation as a variable cost that resets with every new buyer, every new corridor, every new compliance requirement. Each reconstruction is a cost. None accumulates into a reusable asset. The gap between what a large food company can claim and what a smallholder can claim is not a function of product quality. It is a function of documentation infrastructure.
For non-food export sectors encountering the same structural condition: The textile exporter facing Digital Product Passport requirements, the herbal cosmetics producer facing EU Product Information File requirements, the pharmaceutical manufacturer facing FDA raw material origin documentation — each is encountering the same upstream condition through a different regulatory gateway. The documentation architecture in each sector was built for the admissibility question its era required. The intelligibility question now being asked across all of them is structurally identical. The governance gap — no institution owns the connective question — is structurally identical. The condition this paper diagnoses in food systems is not a food problem. It is the condition of every Indian export sector built on producer knowledge that documentation was never designed to carry.
6. WHAT WOULD NEED TO BE TRUE
The structural argument of this paper is not that existing systems have failed. They have not. It is that India's current policy agenda is converging on requirements that no existing system was built to satisfy — and that the structural conditions for satisfying them are identifiable even where the institutional and political decisions about how to satisfy them are not.
The following describes necessary structural conditions — not design prescriptions. The threshold floor established by certification, compliance, and export documentation systems remains essential and is not displaced by any of the conditions below. What is described is the layer above that floor. The conditions are listed in order of dependency: governance assignment is the master condition upon which all others rest.

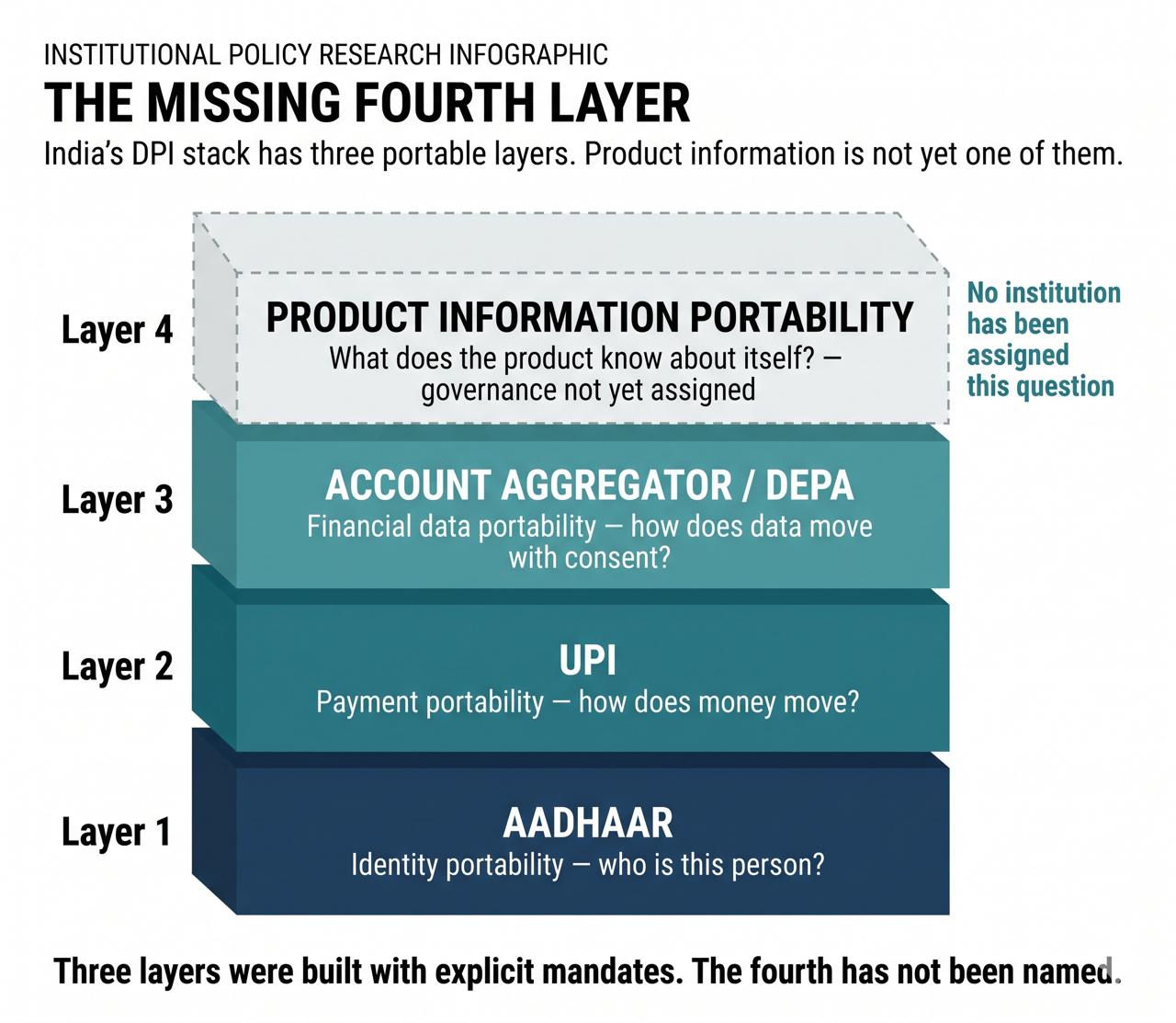
Governance assignment. This is the condition all others depend on. No existing institution currently owns the cross-domain question: how should producer-level product information be structured so that it satisfies FSSAI's requirements, APEDA's documentation standards, international buyer due diligence, and consumer information needs from a single producer declaration? Until that question is assigned to an institution with the mandate and the authority to answer it, the other conditions cannot be met. This paper does not prescribe which institution should hold the assignment. It observes that the absence of such an assignment is what makes the mechanism self-sustaining.
Individual producer resolution. The gap is at the level of what individual producers know about their specific products. An architecture that operates at category, commodity, or certification level replicates the gap rather than closing it. TraceNet's Internal Control System demonstrates that individual-level capture is achievable within grower groups. A generalised architecture would face definitional questions — about the unit of declaration, the treatment of mixed-source products, the handling of producer groups and FPOs — that any governance framework would need to resolve.
Cross-purpose portability. The same producer declaration needs to serve domestic health claim substantiation, export traceability documentation, and buyer due diligence without separate reconstruction for each context. These are different legal categories, and whether a declaration made for one purpose travels to another is a regulatory question requiring explicit governance decisions. The structural observation is that without some form of portability, the reconstruction burden persists: each new context requires information to be reassembled from source rather than carried forward.
Schema specification. For a producer-level layer to feed into FSSAI's requirements, APEDA's documentation standards, and international buyer formats without displacing them, it must use structured formats those systems can receive. Which institutions are authorised to specify receiving formats — and for which downstream contexts — is a governance question the current architecture leaves open.
Source and confidence transparency. A producer-level layer that combines producer-declared attributes with institutionally verified records and third-party test results requires clear attribution for each data type. What counts as adequate evidence for each field, and how producer declarations are distinguished from certified findings, are questions any governance framework would need to address.
The same structural upstream layer that health claim substantiation and trade documentation now require is also the precondition for credible sustainability disclosure — the data that climate and ESG compliance frameworks need shares the same origin in structured, producer-attributed product intelligence, even where the downstream technical schemas differ.
6.1 The Historical Moment
The inherited admissibility architecture is not uniquely Indian. Every trading economy built the same systems under the same constraints. Documentation architectures designed when information moved slowly, was exchanged episodically, and served a smaller number of downstream uses were the right answer for their era. What has changed is not the validity of those systems. It is the speed, frequency, granularity, and multiplicity of uses now expected from product-linked information — across food, textiles, cosmetics, pharmaceuticals, and every other sector in which what a producer knows has acquired measurable downstream value.
The constraints that made the admissibility architecture the only viable option — limited computing capacity, fragmented communication infrastructure, the impossibility of carrying rich structured data across organisational boundaries at reasonable cost — have been removed. What was structurally impractical in 1995 is structurally straightforward in 2026.
Kenya built its National Horticulture Traceability System by embedding farm-level data collection in mandatory export licensing — a governance decision that aligned the incentive to generate information with the institutional purpose it was intended to serve. The result covers 1.2 million smallholders with daily practice logs and real-time data sharing across the export certification ecosystem.ᴷ⁷ The reversal of non-accumulation required one specific institutional decision: this information will be required, this institution is responsible for collecting it, this system will carry it forward.
India has already demonstrated that the architecture works. TraceNet has operated for two decades. The constraint was never technical. The question is not whether a producer-level information layer can be built at scale. The question is what class of institutional decision would need to be made to extend the conditions that made it possible in one corridor to the system as a whole — an institution with cross-domain remit, authority to bind schemas across agencies, and the ability to tie mandates to the corridors where the requirement is most urgent. That decision has not been made.
The millet farmer in Nalgonda still knows more about their crop than any documentation system built to serve them can carry. The processed food manufacturer still assembles the same documentation from scratch for each compliance requirement. The heritage product still reaches commodity markets because no documentation pathway exists to carry its attributes to the buyer who would pay for them. The health claim that is scientifically true still cannot be substantiated at the scale of the producer who could make it.
The paper leaves the governance question open — deliberately, because answering it is an institutional act, not a structural analysis. What the analysis makes clear is that the question has a shape, that India has already shown it can be answered, and that the conditions which made the answer possible once are identifiable, replicable, and have not yet been assigned. The question of what would make an institution choose to own the mandate — what trigger, what incentive, what external pressure, what policy window — is the next question. It is one this paper identifies but does not prescribe. That is the boundary between structural analysis and institutional decision.
REFERENCES
Note on reference numbering: Citations in this paper follow the series-wide numbering system shared across the SGPIS research series. Reference numbers are not sequential within this paper; gaps reflect citations carried by other papers in the series.
-
Government of India. Food Safety and Standards Act, 2006. Ministry of Health and Family Welfare. No. 34 of 2006.
-
Food Safety and Standards Authority of India. Office Order on Implementation of Standardised Format for Submission of Representations Seeking Risk Assessment. File No. SS-G004/1/2024-Standard-FSSAI (Part-I). December 24, 2025. Effective January 1, 2026.
-
Supreme Court of India. 3S And Our Health Society v. Union of India & Anr. W.P.(C) No. 437/2024. Order dated February 10, 2026.
-
European Parliament and Council. Regulation (EU) 2023/1115 on the making available on the Union market of certain commodities associated with deforestation. Official Journal of the European Union, L 150/206, June 2023.
-
European Commission. Commission Implementing Regulation (EU) 2025/1093. Official Journal, May 23, 2025.
-
Field observation, Reverse Buyer-Seller Meet under the RAMP Programme, Hyderabad, April 7–8, 2026. Details on file with the author.
15b. Government of India. Jan Vishwas (Amendment of Provisions) Bill, 2026. Bill No. 104 of 2026. Passed Lok Sabha April 1, 2026; passed Rajya Sabha April 2, 2026.
-
Agricultural and Processed Food Products Export Development Authority. Annual Report 2022–23. Ministry of Commerce and Industry, 2023.
-
Press Information Bureau. Organic Certification under National Programme for Organic Production. July 27, 2025.
-
Agricultural and Processed Food Products Export Development Authority. India Tracenet: Traceability System for Organic and Agricultural Export Products. Ministry of Commerce and Industry.
-
Fripp, E., et al. Traceability and Transparency in Supply Chains for Agricultural and Forest Commodities. Washington DC: WRI and FAO, 2023.
-
Jost, S., Herzig, C., Birringer, M. "A Balancing Act: 20 Years of Nutrition and Health Claims Regulation in Europe." Foods 14, no. 9 (2025): 1651.
-
Taillie, L.S., et al. "Food Industry Compliance With the Display of Front-of-Package Warning Labels at the Final Phase (2020) of Chile's Labeling and Advertising Law." American Journal of Public Health 114, no. 12 (2024).
-
Indian Council of Medical Research – National Institute of Nutrition. Indian Food Composition Tables 2017. Hyderabad: ICMR-NIN, 2017. Also: Abioye V.F., Singh D., et al. "Varietal and processing influence on nutritional and phytochemical composition of finger millet." Heliyon 8 (2022): e11944.
-
Export-Import Bank of India. Enhancing Production and Exports of Millets from India. Mumbai: Exim Bank of India, 2025.
-
Agricultural and Processed Food Products Export Development Authority. Indian Millets. New Delhi: APEDA, accessed April 2026. Also: Food and Agriculture Organization. International Year of Millets 2023. Rome: FAO, 2023.
-
Onmanorama News. "EU deforestation law puts Kerala coffee trade on alert as compliance window narrows." December 4, 2025.
E3. Economic Advisory Council to the Prime Minister. Non-Tariff Barriers Faced by Indian Exporters. EAC-PM/WP/05/2022.
N1. NITI Aayog. Reimagining Agriculture: A Roadmap for Frontier Technology-Led Transformation. New Delhi: NITI Aayog, 2025.
P7. Parliamentary Standing Committee on Communications and IT. Cited in: World Economic Forum, "Digital public infrastructure could transform India's digital agriculture landscape," August 2023.
K7. Feed the Future. "From Plant to Plate: Kenya's National Horticulture Traceability System." Feed the Future, 2024.
-
European Parliament and Council. Regulation (EU) 2024/1781 on the establishment of a framework for setting ecodesign requirements for sustainable products (ESPR). Official Journal of the European Union, L series, June 2024.
-
U.S. Customs and Border Protection. FAQs: Uyghur Forced Labor Prevention Act (UFLPA) Enforcement. Updated 2025. https://www.cbp.gov/trade/forced-labor/faqs-uflpa-enforcement. Cotton and textile detention share derived from CBP UFLPA Statistics Dashboard (2025).
-
European Parliament and Council. Regulation (EC) No 1223/2009 on cosmetic products. Official Journal of the European Union, L 342/59, December 22, 2009. Annex I (Product Information File requirements).
-
International Council for Harmonisation. ICH Q7: Good Manufacturing Practice Guidance for Active Pharmaceutical Ingredients. Adopted by FDA as Guidance for Industry, 2016. Also: U.S. Code of Federal Regulations, Title 21, Part 211 (Current Good Manufacturing Practice for Finished Pharmaceuticals).
-
European Parliament, Panel for the Future of Science and Technology (STOA). Identifying data gaps in the textile industry and assessing current initiatives to address them. EPRS_STU(2024)762850. July 2024.
-
Pawar, P., et al. "Assessment of adulteration in raw herbal trade of important medicinal plants of India using DNA barcoding." 3 Biotech 11 (2021). Also: "Leaf adulteration takes the shine off ashwagandha's gold rush." Vitafoods Insights, 2023.
-
U.S. Food and Drug Administration. Warning Letter: Tyche Industries Ltd. MARCS-CMS 693081. February 6, 2025. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/tyche-industries-ltd-693081-02062025. Supporting: Warning Letter: JHS Svendgaard Hygiene Products Ltd., February 13, 2020.
FURTHER READING — SGPIS SERIES
This paper is the synthesis volume of the Structural Gaps in Product Information Systems series. Earlier papers in the series address specific dimensions of the structural condition diagnosed here:
Ahmed, S. The Limits of Single-Score Nutrition Labels. SGPIS-NL-01. Altibbe Inc., 2026.
Ahmed, S. Certification as Minimum Threshold. SGPIS-GQ-01. Altibbe Inc., 2026.
Ahmed, S. The Information Gap in Food Trade Corridors. SGPIS-TC-01. Altibbe Inc., March 2026.
Ahmed, S. Geographic Indication Without Disclosure Depth. SGPIS-PI-01. Altibbe Inc., April 2026.
All papers are available at altibbe.com/research under CC BY-NC-SA 4.0.
SGPIS-SY-01 | Structural Gaps in Product Information Systems: Synthesis Altibbe Research | April 2026 | California © 2026 Altibbe Inc. This paper is published as independent structural analysis. It does not constitute legal, regulatory, or nutritional advice.

